|
9/7/2023 0 Comments Cobalt 60 nuclear equation Cobalt-60 is used in the inspection of materials to reveal internal structure, flaws, or foreign objects and in the sterilization of food. It is produced by irradiating the stable isotope cobalt-59 with neutrons in a nuclear reactor. It is formed when metal structures, such as steel rods, are exposed to neutron radiation. Cobalt-60 is a byproduct of nuclear reactor operations. For example, cobalt-60 undergoes beta-minus. The most common radioactive isotope of cobalt is cobalt-60 (Co-60). These tests are more sensitive and more accurate if done shortly after exposure. Unlike other types of radioactive decay, gamma decay does not change the nuclear composition of the nuclide. 70 Complete the nuclear equation in wur ansurr booklet or the decav of cesium-137. 
A food irradiation facilitv the cobalt-6(). Bombarding cobalt-59 nuclei With neutrons produces the nuclide cobalt-6(). Notice that the binding energy per nucleon for \(^4He\) is much greater than for the hydrogen isotopes (\(\approx 3 \, MeV/nucleon\)). Cobalt-60 is the longest-lived radioactive isotope of cobalt, with a half-life of 5.27 years. Other techniques that may be used include collecting blood or fecal samples, then measuring the level of cobalt-60. Cobalt.60 is commonlv used as a source of radiation for the prevention of food spoilage. This document is available for review at the Department of Commerce Library, Main Commerce Building, Washington, D.C., under reference number BAW-1130.) is.Their accuracy is limited, of course, by the validity of the assumptions used in setting up and solving the equations, and by the accuracy of the evaluation of the constants. Calculations were then made for the Nuclear Merchant Ship Reactor. Solutions were found for the equations and the constants evaluated from APPR-1 data. The approach was to derive a set of differential equations which should describe the activity buildup phenomenon. How much of a 1.000 mg sample will remain after 15.9 years. Accordingly, the object of this investigation was to determine the concentration of Cobalt allowable as an impurity in the steel which would give no greater activity from Cobalt-60 than from Cobalt-58. Example: The half-life of cobalt-60 is 5.3 years. Practically, there is no object in reducing the Cobalt impurity to a point where the Cobalt-60 activity becomes less than the Cobalt-58 activity. Accordingly, there is some economical limit on the degree of reduction. The greater the reduction in the cobalt impurity, the more expensive the steel becomes. If this is reduced to 0.01%, the Cobalt-60 buildup will be reduced accordingly. Cobalt is similar to iron and nickel in its properties and can be magnetized like iron. The amount of Cobalt in 304 stainless steel varies considerably but is generally on the order of 0.1%. Like a chemical equation, a nuclear equation must be balanced. However, the Cobalt (which is 100% Cobalt-59) is present as an impurity, and it is possible to reduce its concentration considerably. Radioactivity is the decay or disintegration of the nucleus of an atom. In a material such as 304 stainless steel, it is obvious that the nickel cannot be eliminated. Cobalt-58 is formed from the n,p reaction of Nickel-58 while Cobalt-60 is formed from thermal neutron absorption by Cobalt-59. Two of the most active radioisotopes in the deposit are Cobalt-58 and Cobalt-60.  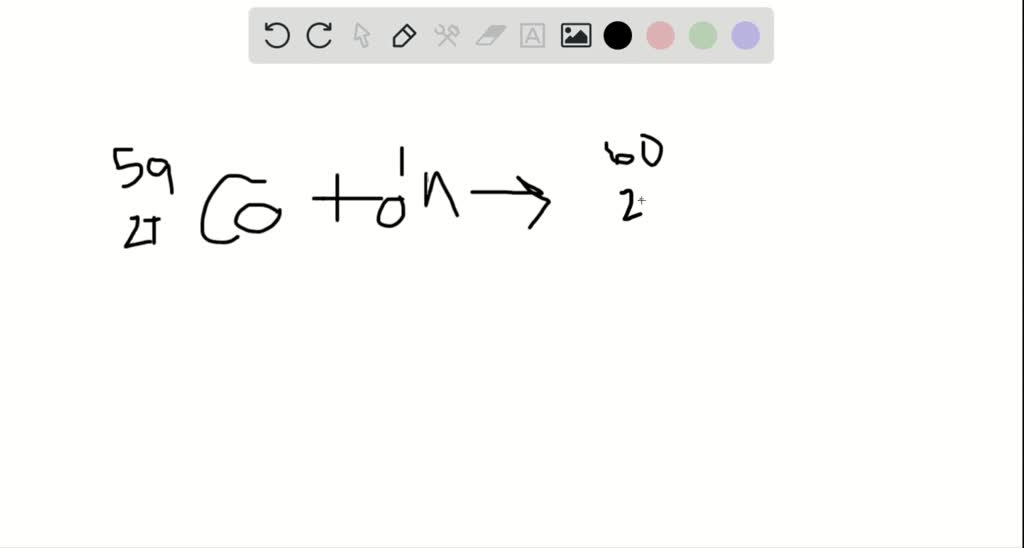
In a given cobalt-60 source, since half of the 2760Co2760Co nuclei decay every 5.27. The nuclear equation for this reaction shows the. This table summarizes the type, nuclear equation, representation. This activity buildup is particularly bothersome during maintenance, i.e. Cobalt-59 is bombarded with neutrons to produce cobalt-60, which is then used to treat certain cancers. In a pressurized water reactor constructed with stainless steel such as the Nuclear Merchant Ship Reactor, a radioactive deposit (commonly called crud) tends to buildup on the walls of the primary system. This reaction has a half-life of about 5. 
COBALT ACTIVITY BUILDUP IN THE NUCLEAR MERCHANT SHIP REACTOR 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
